NON-GMO CERTIFICATION
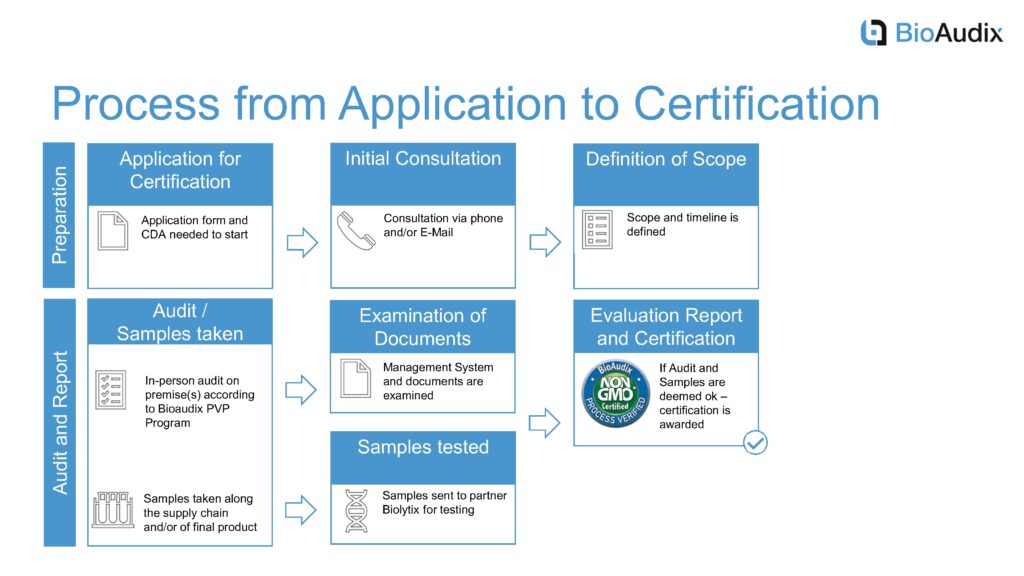
The Bioaudix Non-GMO Standard is dedicated to assist organizations, supplying non-genetically modified product. Applicable to both food and feed, the scope can range from parts of the supply chain to the entire supply chain of the production process.
From the seed, through the growing and harvest, transportation, collection, storing and processing to the market channel, we offer independent certification of the quality management systems.
The EU regulations concerning NON-GMO, including directive 2001/18/EC and regulations 1829/2003 and 1830/2003, governs the labeling requirement of for products with a GMO presence above 0.9%.
The Biolytix standard (a Non-GMO Standard), however, starts where the EU regulation ends, our program certifies Non-GMO products that are below the 0.9% (per cent) threshold. The standard can be applied to all processes in the supply chain.
Critical points of the program include the control of parallel production, product traceability and an effective product recall system. Certification against this standard gives companies the assurance of independent third-party verification by a recognized certification company.
The program is currently being offered to European and other international food manufactures/processors wishing to export non-GMO certified product to the United States and identify their products with the USDA PVP shield. BioAudix intends to expand its services to include US-based companies wishing to market certified non-GMO products under the BioAudix PVP Program in the near future.
Non-GMO supply chain certification does not replace compliance with national and international regulations.
GMO TESTING
Our DNA laboratory of choice, Biolytix (www.biolytix.ch), has been offering GMO testing for over 18 years. Their trained scientists have been involved in the development and validation of protocols for GMO detection used throughout the whole industry. Biolytix testing laboratory operates under strict guidelines and is ISO 17025 accredited. Bioaudix and it’s “NON-GMO Project” is verified by the USDA.
Biolytix uses quantitative PCR-based DNA detection technology to determine the amount of GMO present in samples. It offers a range of GMO detection services that identify presence of GMOs to levels as low as 0.01%.
- Online sample reporting is accessible 24/7 via a password-protected secure website.
- Under the “Processed Verified Program (PVP)” umbrella, BioAudix acts as a certifying agent, providing monitoring and verification of client facilities.
- BioAudix is incorporated owned by three individual investors who are actively involved in the PVP Program.